1. Introduction
Since the 1980s, titanium and its alloys have found many applications in dentistry and orthopedic medicine due to their combination of several characteristics that are very desirable in biomedical applications, including strong corrosion resistance, excellent biocompatibility, low density, and suitable mechanical performance [1, 7, 8, 10, 15, 16]. Due to this combination, titanium-based materials have become particularly suitable to load-bearing applications, including dental prostheses and implants, joint replacements, and other orthopedic applications [7, 8, 11, 15].
Titanium has been of particular interest among the metallic biomaterials due to the generally more favorable biological response of titanium compared to several cobalt- and iron-based alloys. In such materials, elements like Co, Ni and Cr can be emitted in the forms of the ions within the body and will result in untoward biological effects [7, 8, 10, 15, 16]. Commercially pure titanium (CP-Ti) and most titanium alloys on the other hand have demonstrated good biocompatibility both in experimental and clinical practice [5, 7, 8, 10]. This tendency can be explained by the stable passive oxide coating which develops naturally on the surface of titanium and covers the material in physiological conditions [5].
Corrosion resistance is of particular importance to implant materials since the human body is a chemically aggressive environment. In body fluids, chloride ions, organic compounds and the other dissolved species are found and they may speed up the rate of electrochemical degradation [1, 14, 15, 19]. In the case of corrosion, the metal ions and the degradation products can be released into the surrounding tissue, thereby affecting the performance of the implants and increasing the disadvantage of the long-term biological response. For this reason, improving the corrosion resistance of titanium-based alloys remains one of the main goals of biomaterials research [7, 8, 10, 15, 16].
Simultaneously, the corrosion resistance of the implant does not determine its performance alone, but the properties of the surface also play a role. The contact between the implant surface and surrounding hard or soft tissue is the key factor in the process of osseointegration, soft-tissue attachment, and long-term stability [5, 6]. Over the past few years, surface and compositional modifications able to enhance wettability, better tissue response and lessen the risk of bacterial adhesion and peri-implant complications have received a growing interest [5, 6, 9].
Titanium is commercially available in the α-phase form, and it has been utilized successfully in several biomedical applications because of its excellent corrosion resistance and non-toxicity in the human body [1, 7, 10, 15]. Nevertheless, its mechanical characteristics do not always have the required strength and wear resistance as needed by applications [7, 10, 11]. Titanium alloys were developed as a result of this limitation. An example of one of the first and most commonly used is Ti-6Al-4V, providing better strength. Nevertheless, its elastic modulus remains significantly better than the natural bone [10, 11, 15, 16]. Such a discrepancy can lead to stress shielding that can lead to bone resorption and, ultimately, to loosening of the implants.
To address these shortcomings, recent research has been directed at finding new titanium-based biomaterials that have a lower elastic modulus, better corrosion resistance, and non-toxic alloying elements [10, 12, 15, 16, 17]. In this regard, molybdenum is considered an attractive β-stabilizing element to titanium alloys. Ti-Mo systems have been of interest since they could offer a desirable trade-off between mechanical performance and biocompatibility relative to the traditional titanium alloys [10, 13, 16, 19]. However, the behavior of Ti–Mo alloys is strongly influenced by both their composition and the processing conditions. Previous studies have shown that Mo addition can enhance the development of phases like martensitic orthorhombic α″ and ω phases, which can raise the strength but at the same time raise the elastic modulus and and increase brittleness [16].
This is the reason that Ti-Mo alloys still require further modification to enhance their biomedical use. One possible route is the incorporation of ceramic oxides like SiO 2 which could improve the level of corrosion resistance and alter the behavior of surfaces. From this perspective, the current research is an exploration of Ti-15Mo alloy with SiO 2 and its possible usage in biomedical applications especially where better corrosion behavior and good surface characteristics are needed [13, 19].
2. Experimental Work
Ti–15Mo–xSiO₂ specimens were fabricated by powder metallurgy. The average particle size and purity of the starting powders are listed in Table 1. The weighted powders were meticulously mixed in a rotating automatic ball mill using steel balls of different diameters with ethanol serving as the wet medium of mixing. The mixing was conducted during 5 h to obtain a homogenous powder blend. After mixing, 3.5 g of the mixture was then pressed into disk-shaped specimens, 12 mm in diameter and 6 mm in thickness, using an electric hydraulic press. The compaction pressure was 800 Mpa, and the specimens were maintained at the pressure of 800 Mpa in 4 min. The green compacts were then sintered in an electric furnace under an argon atmosphere. The temperature was raised to 950 °C at a heating rate of 10 °C/min and the samples maintained at this temperature for a duration of 7 hours. The specimens were then allowed to cool in the furnace to allow them to attain room temperature after sintering.
Table 1
Characteristics of the powders used in this study
|
Powder |
Average particle size (μm) |
Purity (%) |
|
Ti |
26.43 |
99.85 |
|
Mo |
29.89 |
99.90 |
|
SiO₂ |
53.433 |
99.95 |
2.1 Microstructures Characterization
X-Ray Diffraction. After sintering, The specimens of Ti-based alloys were analyzed via X-ray diffraction (XRD) and the data compared with the reference data. The measurements were conducted by using the Cu ka radiation having a wavelength of 1.54060 Å. The scan range was set at 20°–80° with the step size of 0.02° and the scan speed was 6°/min. The instrument was working at 40 kV and 30 mA.
Microstructure Observation. After sintering, all the specimens were microstructurally examined through sequential grinding followed by 180, 400, 600, 800, 1000, 1200, 1500, and 2000-grit silicon carbide paper. The samples were then polished with diamond paste to obtain a smooth mirror like surface. Etching was performed at room temperature using the solution listed in Table 2. Following etching, they were rinsed in water and dried, and optical microscopy was performed at 400x magnification and scanning electron microscopy (SEM). Phase identification, grain morphology, grain size and grain-boundary features were studied using the microstructural analysis.
Table 2
Composition of the etching solution
|
Constituent |
Amount (mL) |
|
HF |
10 |
|
HNO₃ |
5 |
|
H₂O |
85 |
2.2 Corrosion Test
Polarization testing was used to determine the behavior of corrosion by a standard three electrode electrochemical cell in Ringer solution according to the ASTM standards [18]. The cell included the specimen as the working electrode, platinum as the working electrode, platinum was used as the auxiliary electrode, and a saturated calomel electrode acted as the reference electrode. Winking M Labf200 potentiostat was used to conduct the tests. Potentiometric polarization was performed after stabilization at the open-circuit potential 250 mV below the open-circuit potential to 800 mV above it. The potential was swept in the anodic direction and the resulting current response was recorded. Based on the polarization curves, corrosion potential (E corr ) and corrosion current density (I corr ) were calculated and the corrosion rate was calculated with the help of Equation (1) [18].
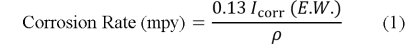
where
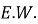
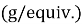
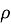
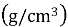
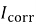
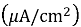
2.3 Contact Angle Test
Contact angle inspection device measure the angle of contact between the liquid (Distilled water) and solid (Ti15Mo-X Sio2) alloys substrate to know the wettability of the electrolyte to the surface of the base sample.
3. Result And Discussion
3.1 Microstructure Characterization
The XRD of the green compact alloys reflect only the starting constituent phases i. e. titanium (Ti), molybdenum (Mo) and silicon oxide (SiO 2 ), as no phase change is experienced during compaction. These transformations are diffusion-controlled reactions and thus need high temperatures to occur. Figure 1 shows the XRD pattern of (Ti-15Mo) alloy with 3 % SiO 2 , which had been sintered at 950 °C during 7 h in an atmosphere of argon. The findings show that following sintering, the initial elemental (Ti) constituents were transformed to two titanium based solid-solution phases, α-Ti and β-Ti. The observed behavior proves that the chosen temperature of sintering and the holding time were adequate to facilitate interdiffusion among the alloying elements and to finalize the phase transformation process.
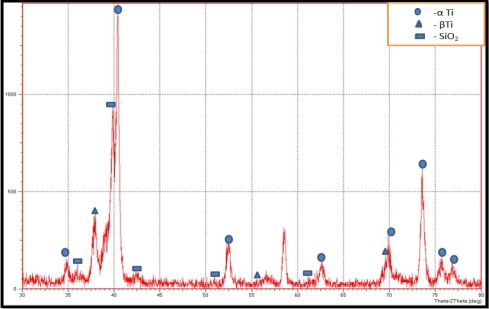
Fig. 1. XRD pattern of Ti–15Mo–3SiO₂ alloy after the sintering process
3.2 Microstructure Representation
The microstructure of the prepared specimens was observed under light optical microscopy (LO). Figure 2 presents optical micrograph of the base Ti-15Mo sintered alloy, as seen under 400x magnification. Within the etched surface, the grain boundaries and the phase contrast in the alloy are easily identified. The microstructure has a duplex morphology made by two different regions as in Figure 2. Brighter areas are the α -Ti phase and the darker areas are the β -Ti phase. This means that the sintered base alloy developed a typical α + β titanium microstructure. The distribution of phases observed also indicates that the powder metallurgy route offered a relatively uniform microstructure across the matrix. This type of microstructural uniformity is significant in that it may lead to predictable material behavior and preferential mechanical performance. Generally, the optical micrograph shows that the two-phase structure as desired in the Ti-15Mo base alloy was achieved successfully after the sintering.

Fig. 2. Optical micrograph of the base Ti–15Mo alloy at 400× magnification
3.3 Contact Angle Test
When the percentage of silicon oxide increases, the contact angle decreases, and this means that the wettability increases, which leads to an increase in the bonding between body tissues and the implanted alloy and reduction of time required for healling. From Table 3 which is shown the variation of contact angles improvement with silicon oxide contents, we notic that there where an increament in improvement percentagre with increasing silicon oxide content, because of silicon oxide addition led to increase surface tension of surface alloy according to the following equation.
Table 3
Contact angle values and percentage reduction for Ti–15Mo–xSiO₂ alloys
|
Sample |
Contact angle (°) |
Reduction (%) |
|
Base |
45.794 |
— |
|
0.5 wt % SiO₂ |
45.657 |
0.30 |
|
1.0 wt % SiO₂ |
42.239 |
7.76 |
|
1.5 wt % SiO₂ |
41.624 |
9.11 |
|
2.0 wt % SiO₂ |
39.528 |
13.68 |
|
2.5 wt % SiO₂ |
38.701 |
15.49 |
|
3.0 wt % SiO₂ |
35.168 |
23.20 |
3.4 Corrosion Test
The electrochemical testing of thecorrosion behavior of Ti–15Mo and Ti–15Mo–xSiO₂ alloys was done in the Hank solution at 37±1°C. The related current-potential relationships, presented as polarization curves in Figure 3 and Figure 4 indicate both the anodic and cathodic responses of the studied alloys. The current dropped with the change in the potential to a minimum value during cathodic polarization. During anodic polarization, the current increased with increasing potential, indicating active anodic dissolution. Meanwhile, the anodic shape also demonstrates that there is a passive region of both the base alloy and the SiO 2 -based alloys, which signifies that a protective surface film forms.
The electrochemical parameters derived from the polarization curves, including the corrosion potential (E corr ), the corrosion current density (I corr ), and the corrosion rate are highlighted in Tables 4. he findings reveal that the corrosion resistance of Ti-15Mo alloy was enhanced by the addition of SiO 2 . In particular, the corrosion current density decreased from 8.619 μA/cm² for the base alloy to 7.8 μA/cm² at 0.5 wt % SiO₂ and further to 1.92 μA/cm² at 3 wt % SiO₂. This reduction in I corr implies that the rate of corrosion decreases hence increased corrosion resistance.
Overall, it is possible to state that the improvement in corrosion performance can be may be associated with the role SiO 2 in promoting to the development of a more protective surface layer that reduces the rate of corrosion of the alloy under the simulated physiologic environment.
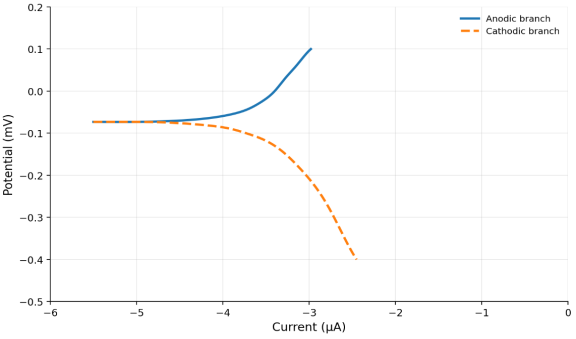
Fig. 3. The corrosion potential Vs Current (μA) for (Ti15Mo) Base Alloy for Hank's solution
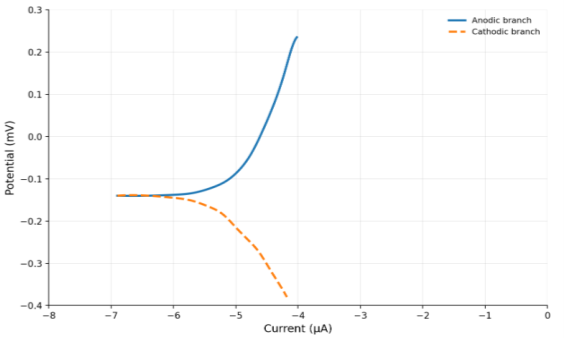
Fig. 4. The corrosion potential Vs Current (μA) for (Ti15Mo-3 %Sio 2 ) Alloy for Hank's solution
Table 4
Porosity, corrosion potential
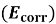
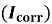
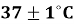
|
wt % |
Porosity(%) |
I corr. (μA/cm 2 ) |
E corr.( mv) |
Corrosion rate (mpy) |
Improvement (%) |
|
0.0 |
18.24 |
8.619 |
-47 |
0.1411 | |
|
0.5 |
20.7 |
7.8 |
-58 |
0.1057 |
25.02 |
|
1.0 |
21.4 |
6.921 |
-136 |
5.49E-09 |
31.65 |
|
1.5 |
23.2 |
5.11 |
-197 |
0.0964 |
54.86 |
|
2.0 |
26.4 |
4.574 |
-50 |
0.0636 |
49.51 |
|
2.5 |
28.6 |
2.54 |
-102 |
0.0712 |
77.49 |
|
3.0 |
30.9 |
1.92 |
-70 |
0.0252 |
82.81 |
4. Conclusion
- Silicon oxide addition enhanced the corrosion resistance of Ti–15Mo alloy. This became even better with increase in the SiO₂ content increased, reaching a maximum of 82.81 % at 3 wt % SiO₂, when the corrosion rate declined to 0.0252 mpy.
- The addition of SiO 2 to Ti-15Mo alloy improved the porosity of the specimen, with the largest porosity of 30.9 %being observed at 3 wt % SiO 2 . This change of composition was associated with better corrosion performance under the current experimental conditions.
- Addition of SiO 2 to decrease the contact angle also was evident, with the highest decrement being observed at 3 wt. SiO 2 . This result indicates improved surface wettability, which was correlated with the increased corrosion behavior of the alloy.
References:
- de Viteri V. S., Fuentes E. Titanium and titanium alloys as biomaterials. Chap. 5 // Tribology — Fundamentals and Advancements / ed. by J. Gegner. — Rijeka: InTech, 2013. — P. 155–181.
- El-Hajje A., Kolos E. C., Wang J. K., Maleksaeedi S., He Z., Wiria F. E. et al. Physical and mechanical characterization of 3D-printed porous titanium for biomedical applications // Journal of Materials Science: Materials in Medicine. — 2014. — Vol. 25, no. 11. — P. 2471–2480.
- Al Deen H. H. J., Hamza S. J. Experimental study of the electrochemical and biological properties of (Nb–1 %Zr–xGe) alloy for biomedical applications // NeuroQuantology. — 2022. — DOI: 10.14704/nq.2022.
- Haleem A. H., Radhi N. S., Jaber N. T., Al-Khafaji Z. Preparation and exploration of nano-multi-layers on 316L stainless steel for surgical tools // Jordan Journal of Mechanical and Industrial Engineering. — 2024. — Vol. 18, no. 2.
- Hanawa T. Biocompatibility of titanium from the viewpoint of its surface // Science and Technology of Advanced Materials. — 2022. — Vol. 23, no. 1. — P. 457–472.
- Hussein E. Y., Al-Murshdy J. M. S., Radhi N. S. Surface improvement of titanium alloys for biomedical applications by anodizing // Jordan Journal of Mechanical and Industrial Engineering. — 2024. — Vol. 18, no. 3.
- Jung H.-D. Titanium and its alloys for biomedical applications // Metals. — 2021. — Vol. 11, no. 12. — Art. 1945.
- Marin E., Lanzutti A. Biomedical applications of titanium alloys: a comprehensive review // Materials. — 2023. — Vol. 17, no. 1. — Art. 114.
- Chakraborty R. et al. MWCNT reinforced bone-like calcium phosphate—hydroxyapatite composite coating developed through pulsed electrodeposition with varying amount of apatite phase and crystallinity to promote superior osteoconduction, cytocompatibility and corrosion protection performance compared to bare metallic implant surface // Surface and Coatings Technology. — 2017. — Vol. 325. — P. 496–514.
- Li Y. et al. New developments of Ti-based alloys for biomedical applications // Materials. — 2014. — Vol. 7, no. 3. — P. 1709–1800.
- Long M., Rack H. J. Titanium alloys in total joint replacement — a materials science perspective // Biomaterials. — 1998. — Vol. 19, no. 18. — P. 1621–1639.
- Hanawa T. Recent development of new alloys for biomedical use // Materials Science Forum. — 2006. — Vol. 512.
- Abdul-husien M., Al Deen H. H. J., Al-Roubai A. O. Improvement of corrosion and wear resistance of (Ti12Mo5Ta) alloy by germanium addition used in biomedical applications // Jordan Journal of Mechanical and Industrial Engineering. — 2024. — Vol. 7, no. 1.
- More N. S. et al. Tribocorrosion behavior of β titanium alloys in physiological solutions containing synovial components // Materials Science and Engineering: C. — 2011. — Vol. 31, no. 2. — P. 400–408.
- Geetha M. et al. Ti based biomaterials, the ultimate choice for orthopaedic implants — a review // Progress in Materials Science. — 2009. — Vol. 54, no. 3. — P. 397–425.
- Niinomi M., Nakai M., Hieda J. Development of new metallic alloys for biomedical applications // Acta Biomaterialia. — 2012. — Vol. 8, no. 11. — P. 3888–3903.
- Ikeda M., Ueda M., Niinomi M. Recent studies and developments in titanium biomaterials // MATEC Web of Conferences. — 2020. — Vol. 321.
- ASTM. Standard practice for microetching metals and alloys. — United States of America: ASTM, 2005.
- Şimşek I., Özyürek D. Investigation of wear and corrosion behaviors of Ti15Mo alloy produced by mechanical alloying method in SBF environment // Powder Metallurgy and Metal Ceramics. — 2019. — Vol. 58, no. 7. — P. 446–454.

